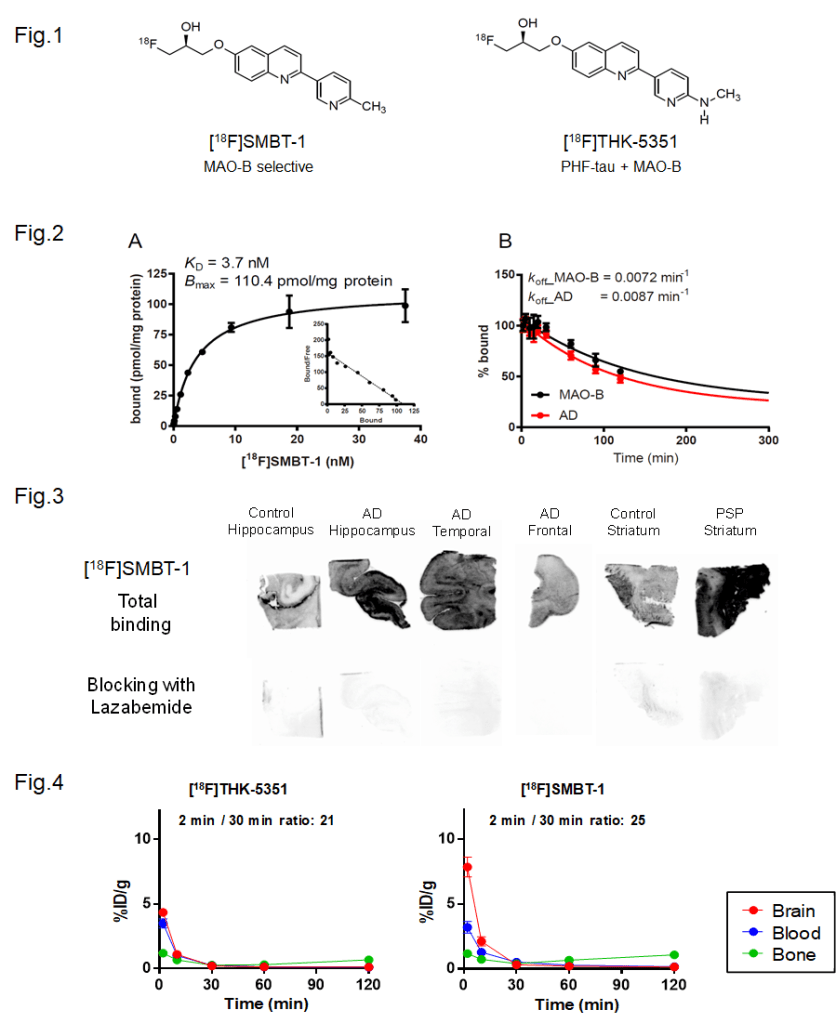
[18F]SMBT-1 (Fig. 1) was developed as a selective and reversible monoamine oxidase B (MAO-B) PET tracer derived through the optimization of [18F]THK-5351, which was known as first-generation tau PET tracer (Okamura et al. Clin Transl Imaging (2018)).
As shown in Figure 2, SMBT-1 showed strong and reversible binding to MAO-B (Kd = 3.7 nM), and low binding affinity to other enzymes, receptors and misfolded proteins such as amyloid-β and tau. In the autoradiography using human brain sections, greater amount of [18F]SMBT-1 binding was observed in the brain sections from patients with Alzheimer’s disease (AD) and progressive supranuclear palsy (PSP) than in control brain sections. These [18F]SMBT-1 bindings were completely displaced with MAO-B inhibitor lazabemide (Fig. 3), suggesting high binding selectivity of this tracer to MAO-B. Furthermore, [18F]SMBT-1 showed an excellent pharmacokinetics in normal mice (Fig. 4) (Harada et al. J Nucl Med (2021)).
In First-in-human PET studies performed in Austin Health, Melbourne, [18F]SMBT-1 yielded high contrast images at 60-90 min post injection. In AD patients, [18F]SMBT-1 retention was higher in the neocortex. More than 85% of [18F]SMBT-1 signal was blocked and no residual cortical activity was observed after the selegiline regimen, indicating high selectivity for MAO-B (Villemagne et al. J Nucl Med (2021)).